This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. James Chadwick, in full Sir James Chadwick, (born October 20, 1891, Manchester, England-died July 24, 1974, Cambridge, Cambridgeshire), English physicist who received the Nobel Prize for Physics in 1935 for the discovery of the neutron. The neutron was discovered in 1932, when James Chadwick used scattering data to calculate the mass of the neutral particle.How did sir James chadwick discover the neutron?Who was James Chadwick? He was studying radioactivity at the time with Ernest Rutherford. The neutron was discovered in 1932, when James Chadwick used scattering data to calculate the mass of the neutral particle. After Chadwick’s experiment, Werner Heisenberg proposed that the nucleus is made up of protons and neutrons, which was later found to be true. This “radiation” turned out to be a stream of neutrons. When this occurred, highly penetrating radiation was emitted. 
How did James Chadwick discover the neutron?Ĭhadwick found the neutron by hitting a chemical called beryllium with alpha particles. Jame’s Chadwick discovery of the neutron is present in the modern atomic model. Point out which contributions are present in the modern atomic model and which were eventually disproven and thus are not part of the modern model. Which of James Chadwick’s contributions are present in the modern atomic model and which were eventually disproven and thus are not part of the modern model? Chadwick’s discovery forced a revision of the cloud model, and scientists sometimes refer to the revised version as the James Chadwick atomic model. 
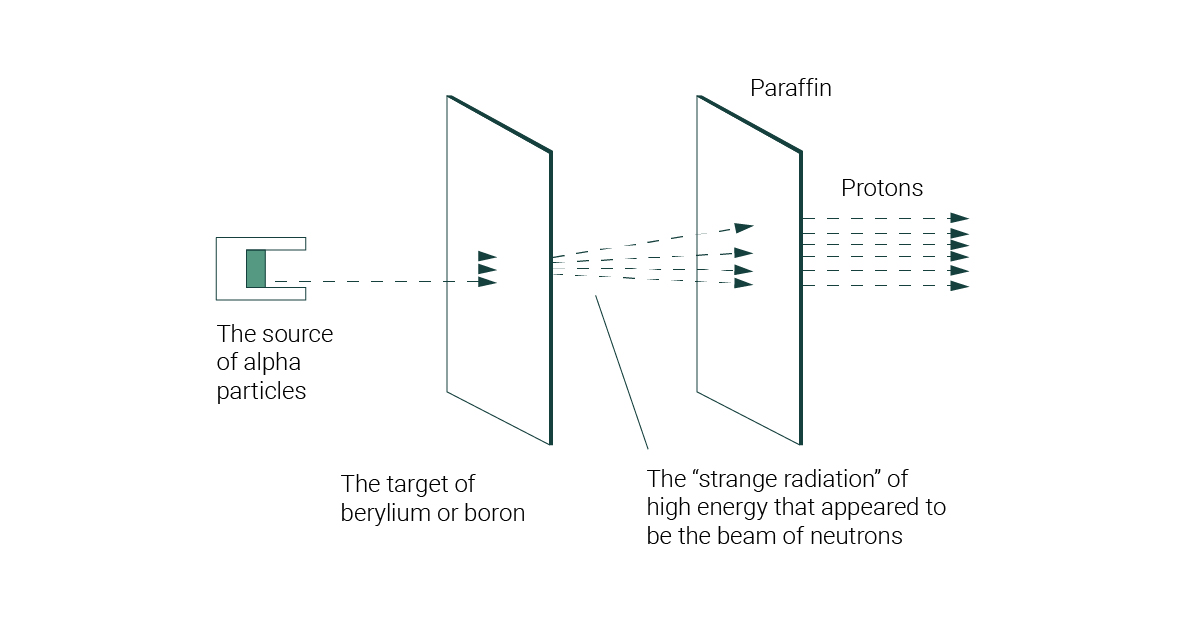
What model did James Chadwick create?Ĭhadwick is responsible for discovering the existence of the neutron, the neutral particle that shares the nucleus with the positively charged proton. In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron. What did Chadwick discover about atoms?īy 1920, physicists knew that most of the mass of the atom was located in a nucleus at its center, and that this central core contained protons. In 1932, Chadwick made a fundamental discovery in the domain of nuclear science: he proved the existence of neutrons – elementary particles devoid of any electrical charge. What was James Chadwick main contribution to the atomic theory? 3 What model did James Chadwick create?.2 What did Chadwick discover about atoms?.1 What was James Chadwick main contribution to the atomic theory?.With Chadwick's announcement, Heisenberg then proposed the proton-neutron model for the nucleus. In 1935, he was awarded the Nobel Prize for his discovery. In 1932, Chadwick proposed that this particle was Rutherford's neutron. This stream was originally thought to be gamma radiation.įurther investigations into the properties of this radiation revealed contradictory results. Like gamma rays, these rays were extremely penetrating and, since they were not deflected upon passing through a magnetic field, neutral. However, unlike gamma rays, these rays did not discharge charged electroscopes (the photoelectric effect). In 1930 it was discovered that Beryllium, when bombarded by alpha particles, emitted a very energetic stream of radiation. According to PhysicsLab Online, James Chadwick was assigned the task of tracking down evidence of Rutherford's tightly bound, but theoretical, "proton-electron pair." Chadwick's experiment showed this was actually a different subatomic particle, now called the neutron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
